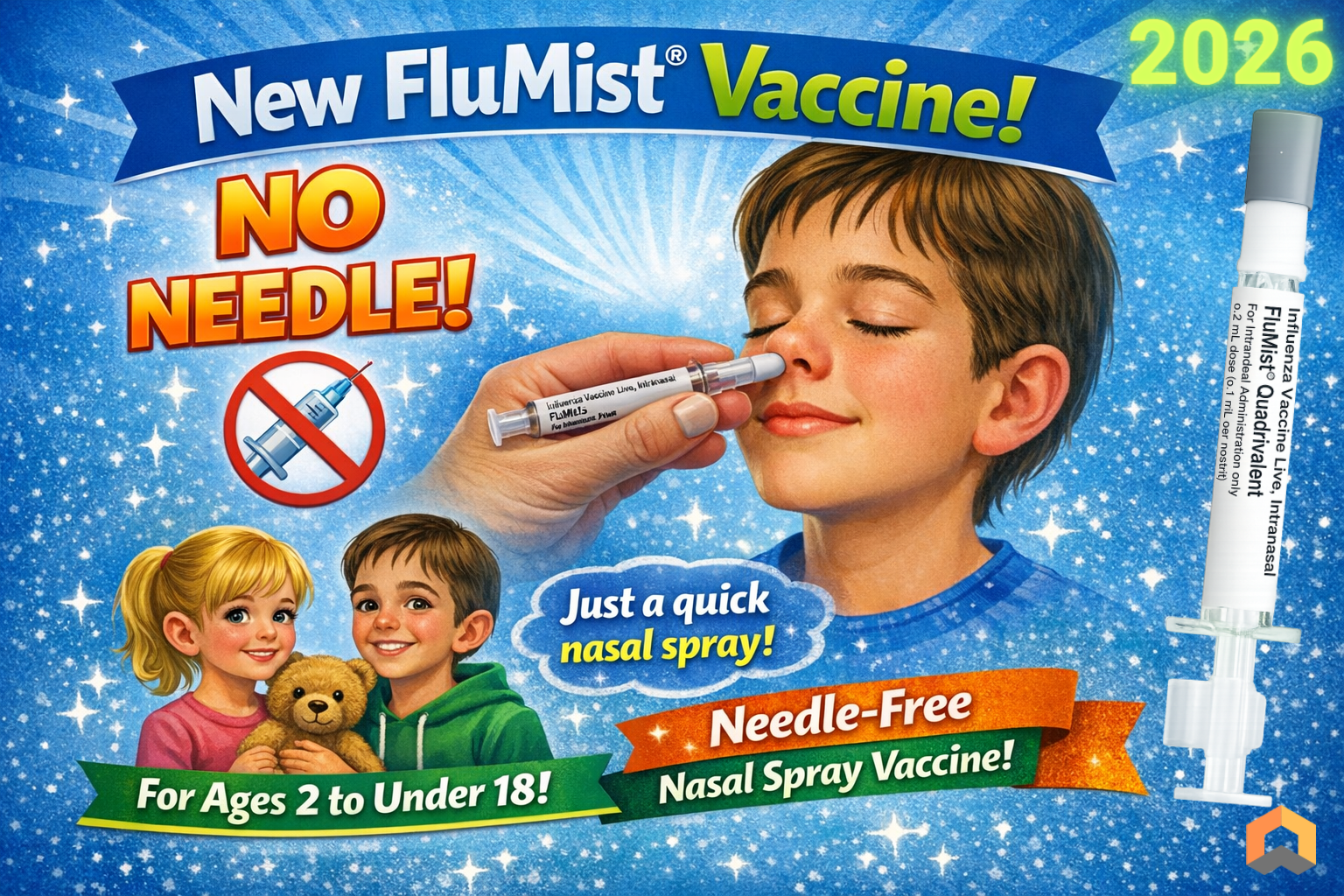
No needle
From 2 years to under 18 years
Contraindications:
Severe immunocompromised or on long term aspirin
2-4 years old children with wheeze or asthma within the past 12 months
Runny or severe blocked nose
No needle
From 2 years to under 18 years
Contraindications:
Severe immunocompromised or on long term aspirin
2-4 years old children with wheeze or asthma within the past 12 months
Runny or severe blocked nose

Standard consultation and chronic disease care plan items (3, 23, 36, 44, 2715, 2717, 965, 967) will be bulk-billed – applicable to prebooked and on-the-day appointments, on weekdays and weekends.
Procedures and specialised services will continue to attract gap fees, including 24-hour ambulatory blood pressure monitoring, skin excisions & biopsies, Implanon insertion/removal, and other procedural items.
Gap fees may vary depending on each doctor’s individual fee schedule.
These billing changes aim to ease patients’ expenses while enabling our clinic to maintain service quality and meet rising operating costs.
If you are unsure whether your appointment will be bulk-billed or privately billed, please contact reception.
The National Lung Cancer Screening Program (NLCSP) uses low-dose CT scans every 2 years to look for lung cancer in high-risk people without any symptoms. It aims to find lung cancer early and save lives. Screening for eligible people starts from July 2025.
Please book an appointment with your doctor for more information.
Eligibility:
- Between 50 and 70 years and
- show no signs or symptoms and
- currently smoke or have quit smoking in the past 10 years and
- have a history of tobacco smoking of at least 30 pack-years.
Definition of ‘pack-years’
The term ‘pack-year’ is a way of measuring the number of cigarettes a person has smoked in their lifetime.
Pack-years are calculated by multiplying the number of cigarette packs smoked per day by the number of years the person has smoked.


• RSV is a common virus that easily spreads.
• In Australia, RSV infection is the most common cause of infant hospitalisation because it can often progress to pneumonia (infection of the lungs) or bronchiolitis (inflammation of the small airways in the lungs) in very young children.
• Eligible women from 28 to 36 weeks of pregnancy will be able to access free Abrysvo® RSV vaccine through the National Immunisation Program (NIP)
• When the Abrysvo® RSV vaccine is given in pregnancy, the mother will create protective antibodies and pass these to their baby through the placenta. This helps to protect their newborn baby from birth and in its first few months of life. Immunisation in pregnancy reduces the risk of severe RSV illness in infants under 6 months of age by around 70%.
• Victoria will fund a long-acting RSV monoclonal antibody, nirsevimab (Beyfortus™) for newborns and infants whose mothers did not receive Abrysvo® RSV vaccine. Nirsevimab for infants will be available between 1 April 2025 to 30 September 2025.
• 75 years and older: Recommended every 6 months.
• 65-74 years: Recommended every 12 months
• 18-64 years:
◦ Without severe immunocompromise: Can receive every 12 months.
◦ With severe immunocompromise: Recommended every 12 months
• 5-17 years
◦ Without severe immunocompromise: Not recommended.
◦ With severe immunocompromise: Can receive every 12 months.
• Under 5 years: Not recommended.

From November 2023, the shingles vaccine Shingrix® will be available on the National Immunisation Program (NIP) schedule for the prevention of shingles and post-herpetic neuralgia.
A 2-dose course of Shingrix® (usual cost $560) will be available for free for:
People aged 65 years and older
First Nations people aged 50 years and older
Immunocompromised people aged 18 years and older with the following medical conditions:
haemopoietic stem cell transplant
solid organ transplant
haematological malignancy
advanced or untreated HIV.

MyMedicare is a new voluntary patient registration system.
It aims to strengthen the relationship between patients and general practice. MyMedicare patients will have access to longer funded telehealth consultations with your GP and other benefits which will be clarified in the future.
You’re eligible to register Main Road Medical as your regular clinic if you have:
A Medicare card or DVA Card
Two face-to-face visits at Main Road Medical in the previous 24 months.
People experiencing domestic violence and homelessness will be exempt from eligibility requirement.
Parents/guardians and children can be registered at the same practice if one of them is eligible and registered.
Young people aged 14 to 17 years can register and provide consent without a parent/guardian.
To register with MyMedicare please follow the below steps
Install the Express Plus Medicare Mobile app on you smart phone
Select Main Road Medical as your regular practice (please note our practice name includes no “Centre”)
Select your preferred GP
Submit your registration

• Invasive Meningococcal Disease is a rare but very serious disease that can cause significant illness, disability and death (in 5 to 10% of cases).
• The Meningococcal ACWY vaccine has been included on the National Immunisation Program for children aged 12 months of age since July 2018.
• MenB has become the most common strain over the past 3 years. However Meningococcal serogroup B vaccine is currently not included in the National Immunisation Program schedule (except in South Australia).
WHO SHOULD BE VACCINATED AGAINST MENINGOCOCCAL B
• Infants, children and adolescents
• Young adults who are smokers or live in ‘close quarters’ including new military recruits and students in residential accommodation.
• Free under National Immunisation Program for people of all ages with asplenia and hyposplenia, complement deficiency and those receiving treatment with eculizumab.
Whooping cough is extremely contagious. It is particularly serious in babies under six months of age, who are at risk of severe complications.
Vaccination during pregnancy (between 20 and 32 weeks gestation) reduces the risk of whooping cough in young infants by 90%
Pregnant women are offered free pertussis-containing vaccine (funded under the National Immunisation Program).
However, the Victorian Government funded pertussis vaccination program for partners of pregnant women and guardians of infants under 6 months of age (if they had not received a pertussis vaccine in the past 10 years) will cease on 30 June 2023. They are still strongly recommended to have a pertussis booster.

Talk to your Doctor about shingles vaccines
About 1 in 3 people will develop shingles in their lifetime, regardless of how healthy they may feel.
Anyone who has had chickenpox can develop shingles, especially people over 50, those with a weakened immune system, and those who had chickenpox in their first year of life.
Nearly all adults 50 years and older already carry the inactive virus that causes shingles.
The likelihood of painful complications from shingles increases with age.

Paxlovid and Molnupiravir — two oral antivirals for COVID-19 infection — are effective in preventing severe disease, hospitalisation and death.
On 1st April, the federal government expanded the Pharmaceutical Benefits Scheme (PBS) eligibility criteria to subsidised COVID-19 antiviral treatments as below.
70 years of age or older, regardless of risk factors and with or without symptoms.
60 years of age or older with at least one risk factor
living in residential aged care
living with disability with multiple conditions and/or frailty
neurological conditions like stroke or dementia and demyelinating conditions e.g. multiple sclerosis, Guillain-Barre Syndrome
chronic respiratory conditions including COPD, moderate or severe asthma
obesity or diabetes (type I or II requiring medication)
heart failure, coronary artery disease, cardiomyopathies
kidney failure or liver cirrhosis
living remotely with reduced access to higher level healthcare
past COVID-19 infection episode resulting in hospitalisation.
ATAGI recommends a 2023 COVID-19 vaccine booster dose for adults in the following groups, if their last COVID-19 vaccine dose or confirmed infection was 6 months ago or longer.


From Monday 11/07/2022 there will be changes to the eligibility criteria of the oral treatments for COVID-19 recommended by the Pharmaceutical Benefits Advisory Committee.
If you are tested positive for COVID-19 by PCR or medically supervised RAT, you may be eligible for antivirals treatment if you are
From 70 years of age
From 50 years of age with 2 risk factors
From 30 years of age with 2 risk factors if Aboriginal or Torres Strait Islander
The risk factors include
living in residential aged care
living with disability with multiple conditions and/or frailty (but not limited to living in supported accommodation)
neurological conditions like stroke or dementia and demyelinating conditions e.g. multiple sclerosis, Guillain-Barre Syndrome
chronic respiratory conditions including COPD, moderate or severe asthma
obesity or diabetes (type I or II requiring medication)
heart failure, coronary artery disease, cardiomyopathies
kidney failure or liver cirrhosis
living remotely with reduced access to higher level healthcare

The pandemic is NOT over yet.
In the last 24 hours, Australia recorded 39,000 cases and 59 death. The third Omicron wave is coming.
The Australian Technical Advisory Group on Immunisation (ATAGI) just updated its recommendations:
From Monday, 11 July 2022, people aged 50 to 64 years should have their fourth dose, while people aged 30 to 49 years may choose to have a fourth shot.
ATAGI also recommended the interval between vaccine doses, or prior infection (whichever comes later), be reduced from four months to three months.
24-hour ambulatory BP monitoring - the gold standard method to assess blood pressure - is now available at Main Road Medical.
Please make an appointment with your doctor to discuss about this investigation.
To be eligible for Medicare rebates, the patient must have not commenced anti‑hypertensive therapy and must meet one of the following criteria
Clinic systolic blood pressure >= 140 mmHg and <= 180 mmHg
Clinic diastolic blood pressure >= 90 mmHg and <= 110 mmHg
To do this test, you will need to wear a blood pressure unit. The unit consists of a blood pressure cuff that is worn on your upper arm and connected to a monitor. You wear the monitor on a strap over your shoulder.
For the next 24 hours, your heart rate and blood pressure will be measured at 20-30 minute intervals during the day and night. When you feel the cuff getting tighter, it’s important to stop and rest until the measurement is complete. Usually each reading takes 30-40 seconds.
It will take about 15 minutes to have the cuff put on and get instructions in the clinic. You will need to return to the clinic at the same time the next day (24 hours later) to have the strap and monitor taken off.
While you are wearing the monitoring unit you must:
Wear a loose-fitting shirt
Remain still during the readings (when the cuff tightens)
Record the time, any symptoms, and your mood, activity, and bedtime in your patient diary
Make sure the monitor stays connected while you undress
Take your normal medications.
You must also follow these important guidelines:
Don’t wear long-sleeved sweaters or dresses
Don’t take the cuff off or remove the batteries from the unit
Don’t get the monitor wet (i.e., don’t swim, shower or bathe)
Don’t drive or operate machinery or use power tools or heavy equipment, as vibrations can disrupt the monitor.

The Australian Technical Advisory Group on Immunisation (ATAGI) has recommended that more people from 16 to 64 years old be given a fourth ‘winter’ dose of COVID-19 vaccine.
Cancer
Chronic inflammatory conditions: Systemic lupus erythematosus, rheumatoid arthritis, Crohn’s disease, ulcerative colitis, …
Chronic lung disease: including severe asthma (requiring frequent hospital visits or the use of multiple medications).
Chronic liver disease
Severe chronic kidney disease (stage 4 or 5)
Chronic neurological disease: stroke, dementia, epilepsy, …
Diabetes mellitus requiring medication
Chronic cardiac disease
People with disability with significant or complex health needs
Severe obesity with BMI ≥ 40 kg/m2
Severe underweight with BMI < 16.5 kg/m2
Annual influenza vaccination is free under the National Immunisation Program for
Children from 6 months to less than 5 years of age
Adults aged 65 years and over
Aboriginal and Torres Strait Islander people aged 6 months and over
Pregnant women (during any stage of pregnancy)
People aged 6 months and over with medical conditions which increase the risk of complications.
Cardiac disease
Chronic respiratory conditions
Chronic neurological conditions
Immunocompromising conditions
Diabetes and other metabolic disorders
Renal disease
Haematological disorders
Children aged six months to 10 years on long term aspirin therapy.
Children under nine years receiving their influenza vaccination for the first time require two doses of vaccine, spaced by a minimum of one month.
Today ATAGI recommends an additional booster dose of COVID-19 vaccine for selected groups who are at greatest risk of severe illness from COVID-19. These groups are:
Adults aged 65 years and older
Residents of aged care or disability care facilities
People aged 16 years and older with severe immunocompromise (as defined in the ATAGI statement on the use of a 3rd primary dose of COVID-19 vaccine in individuals who are severely immunocompromised)
Aboriginal and Torres Strait Islander people aged 50 years and older.
The additional winter booster dose can be given from 4 months after the first booster dose, or from 4 months after a confirmed SARS-CoV-2 infection, if infection occurred since the person’s first COVID-19 booster dose.
Influenza vaccine can be co-administered with the additional booster dose of COVID-19 vaccine.
Comirnaty (Pfizer) or Spikevax (Moderna) are the preferred vaccines for COVID-19 booster doses.
Vaxzevria (AstraZeneca) can be used when an mRNA vaccine is contraindicated or a person declines vaccination with an mRNA vaccine.
Nuvaxovid (Novavax) can be used if no other COVID-19 vaccine is considered suitable for that person.